For several days in late March, North Macedonian citizen Rodzer Zekirovski had all the symptoms of COVID-19: a high fever, aching muscles, and a dry cough. But he couldn’t manage to get a lab test at a state hospital.
Instead, Zekirovski went for what seemed like a good alternative: a Dutch-made test — advertised by a private clinic as high quality — that could accurately detect the disease in mere minutes. Or at least that’s what he thought as he headed home from the clinic, relieved, with a negative test result.
Three days later, Zekirovski, 45, was dead. The official results from a subsequent test showed he had indeed been infected with SARS-CoV-2, the virus that causes COVID-19.
Zekirovski’s wife, Gjultena, said the false negative led his family to take risks that were, in hindsight, terrifying.
“When he tested negative, we were all so happy,” she said. “We were hugging and kissing, children and parents.”
“How could we know?”
While it’s impossible to say whether Zekirovski would have survived if he had known he had COVID-19, his death is a striking illustration of the pitfalls of a tool many have hoped would be a key weapon in the fight against the pandemic: rapid antibody tests.
Unlike molecular tests, which detect proteins of the SARS-CoV-2 virus, and can take hours or days, these rapid tests look for antibodies produced by the immune system as it fights the virus. Results are typically ready in under 30 minutes.
But an investigation by OCCRP and partners has uncovered serious problems with the global market for rapid tests.
Nothing Zekirovski was led to believe about the test he took was true. It never should have been offered as a conclusive diagnostic tool, and it was not nearly as accurate as advertised.
It also wasn’t really made in the Netherlands, reporters have found.
Instead, the test — sold by Dutch company Inzek International Trading under the brand name Biozek — was one of at least three brands of rapid coronavirus tests released by European and U.S. manufacturers that are in fact relabelings of a popular Chinese model.
The test, manufactured by Hangzhou Alltest Biotech Co. Ltd, has been found in some independent trials to be less accurate than claimed. The UK government reportedly canceled a multi-million-dollar order for Alltest kits after a review led by Oxford University found that Alltest and another Chinese brand failed to meet basic standards for accuracy.
And yet the tests are being sold by the million around the world — often promoted as a quality European product, in implicit contrast to less prestigious Chinese manufactures.
The same Chinese tests, under different brand names, have been used by health services and government authorities in countries including Spain, Italy, Great Britain, Indonesia, Russia, and the Vatican. Some have ended up in the hands of online retailers that sell directly to non-healthcare professionals, a practice that violates safe use guidelines. They have even been smuggled into Ukraine in a shipment of clothing.
Buying and relabeling such tests is legal in the U.S. and in European Union countries. Misleading consumers and patients, however, is not.
“A False Negative Is A Disaster”
Rapid tests typically work by taking a tiny sample of blood and placing it on a small plastic strip to check for the presence of viral antibodies. Most tests look for two kinds of antibodies: Immunoglobulin M (IgM), which typically shows up in the earlier stages of inflection, and Immunoglobulin G (IgG), which shows up later and persists longer.
The World Health Organization (WHO) currently does not advise using them for patient care, instead encouraging more research into the tests' effectiveness as a tool for tracking community spread.
Along with ventilators and masks, rapid tests became an in-demand item in the frenzied early weeks of the pandemic. The test kits formed a much-publicized part of Chinese aid shipments and sales to European countries stricken by the virus, but were soon plagued by controversy. Most famously, Spain returned a batch of kits to a Chinese manufacturer, Shenzhen Bioeasy Biotechnology, after they were found to be wildly inaccurate.
On paper, the Alltest brand is far more precise. The company, based in the eastern Chinese city of Hangzhou, is a longtime manufacturer of strip testing kits that detect common infections, drugs, and pregnancy.
Alltest released a small clinical study claiming that its tests are 92.9 percent accurate in detecting IgM and 98.6 percent accurate for IgG.
But independent trials have come up with far weaker results. Crucially, the tests were found to be far less effective for patients who are in the first weeks of the infection. A non-peer-reviewed study of nine commercially available rapid tests released in early April ranked Alltest as the least effective. A separate Spanish study found the Alltest gave false negatives more than half the time.
One of the authors of the study, Juan Cuadros Gonzalez, told OCCRP that the test becomes much more accurate four weeks after the onset of COVID-19 symptoms, with the rate of error dropping to under five percent.
Knowing these limitations, Cuadros and his colleagues have been able to use the tests as a complementary screening tool alongside molecular lab tests. But this one-month lag “is not indicated on the package leaflet, and therein lies the problem,” he said.
Marien de Jonge, a scientist involved in COVID-19 research at the Netherland’s Radboud University Medical Center, said false negatives can be very dangerous.
“When you want to determine if someone is infected, a false positive result is not a big problem, because the only consequence is that someone shuts himself in for nothing,” de Jonge said.
“But a false negative is a disaster,” he said, because wrongly giving someone an all-clear can endanger others in the community.
The clinical trial results released by Alltest claiming such a high level of accuracy are “rubbish,” Claire Standley, an assistant research professor at Georgetown University's Center for Global Health Science and Security, told OCCRP after reviewing the document.
“There’s no information on the clinical state of the patients,” Standley said. “We don’t know if they were hospitalized, we don’t know if they were randomly picked from the community, we don’t know if they had mild symptoms, we don’t know how they were identified. There’s no information about the ethical approvals that were granted to ensure they were consenting to be part of the study. These are all fundamental aspects.”
Clinical trials of rapid tests should compare results obtained from the kits with more accurate lab tests, Standley said. The clinical trial, however, includes no such information, and gives no indication for where to find it.
This doesn’t mean the tests are useless. The Alltest could still be a useful screening tool in tandem with molecular testing, Standley said. It could also be useful as a means of conducting community-wide surveys of the spread of the disease.
Even Alltest states publicly that this is the proper use for its test. In a statement released by the company in response to reports about the cancelled British order, the company said its product is “not suitable for early screening” and should be used as a “supplementary method” to other tests.
Alltest declined to respond to written questions from OCCRP.
“Maybe There is One Word Wrong”
At least three other brands based in Europe and the United States are currently selling Alltest’s models under their own labels, reporters found.
OCCRP reporters were able to track down the tests by searching for key figures from Alltest’s original clinical study, as well as the test model’s reference number, INCP-402, and variations used by Alltest and its clients. That information was also cross-checked with company registration documents and information from ImportGenius, a commercial import-export database.
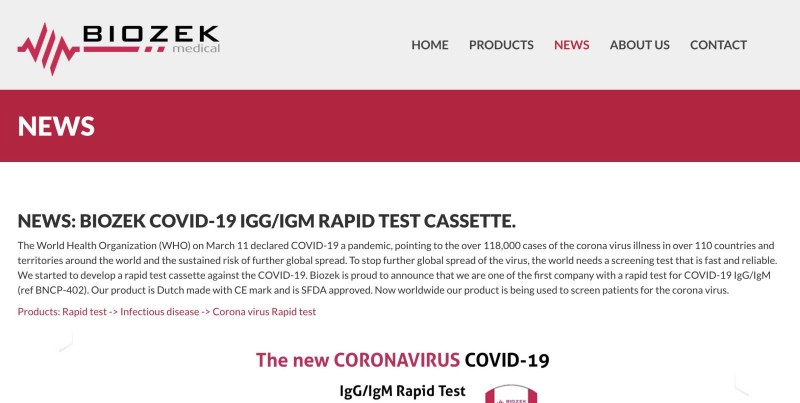
The most prominent relabeler has been the manufacturer of the Biozek test, Inzek International Trading BV, which is located in the Dutch city of Apeldoorn.
Until recently, the company advertised its coronavirus tests on its webpage as Dutch-made and emblazoned them with the “CE” marking, indicating they are made according to European Economic Area standards.
The claim of being Dutch-made has been a key draw for some buyers. On April 8, Indonesian state pharmaceutical company PT Kimia Farma announced the purchase of 300,000 of Biozek’s test kits, touting them as having been “developed under EU regulations.”
Mustafa Hamid, the quality control manager of Biozek, was quoted in the same release as saying, “The tests have been developed and produced under very strict EU and Dutch regulations. These regulations leave no room for error and thus makes for a very reliable and safe product.”
But in fact, the tests were developed and produced in China by Alltest, Inzek CEO Zeki Hamid admitted when questioned by a reporter.
He said the Dutch company would typically conduct its own quality checks in Iraq and carry out some assembly in the Netherlands, but that neither of those steps were possible for the COVID-19 antibody tests due to lockdown measures.
“The party in China is a really professional manufacturer and they have official certificates,” Hamid said. “We trust them one hundred percent.”
Asked why the tests were promoted as Dutch-made on the website, Hamid said, “Maybe there is one word wrong. Maybe we should correct that.” The website was changed the following day, April 29, to remove the claim.
Andi Prazos, a director at Indonesia’s Kimia Farma, said the state company had been convinced the tests were Dutch made. Kimia staff were even given a tour of Inzek’s production facility in the Netherlands before placing their order, he said.
“Before the transaction, we made sure that it’s a credible company and has its own production line. The Indonesian Embassy in the Netherlands also helped us,” Prazos said. The tests have already been distributed to 14 of Indonesia’s provinces, he added.
In response to reporters’ questions, the Netherlands’ Foundation for the Code of Conduct for Medical Devices said it was unable to comment on particular cases but that “parties affiliated with the [foundation] obviously do not consider it desirable if medical devices are advertised in a manner that is incorrect, misleading and unsubstantiated.” Complaints would have to be addressed by a civil court or the country’s Advertising Code Committee, it said.
“It’s still sneaky, clearly, it’s not very transparent to the consumer,” Georgetown’s Standley said, “but it may be [legally] above board and just one of those practices that we didn’t know about because we’ve never been this interested in diagnostics until now.”
Inzek’s “Dutch-made” tests have proved popular. According to Hamid, the company has sold and shipped 1.6 million test kits around the world. The company’s largest customers are in Indonesia, Russia, the Netherlands, Kuwait, Saudi Arabia, and Iraq, he said.
Even the Holy See ordered 700 Biozek tests, Italian newspaper La Stampa reported. The deal was arranged with the help of Paolo Zampolli, a New York-based Italian businessman who reportedly organized a party in 1998 where future U.S. President Donald J. Trump met his wife, Melania Knauss. Zampolli memorialized his role in the Biozek test deal with a photograph on his Instagram page.
“We Do Not Recognize Them as Valid”
By comparing test reference numbers and antibody detection figures, reporters were able to find at least two other mass-marketed rebrandings of the Alltest kits.
One is sold under the house brand name of ScreenItalia, a test manufacturer in the Italian region of Umbria.
The company’s own website includes a declaration of EC conformity in which the Italian company lists itself as the manufacturer of three rapid coronavirus tests bearing Alltest reference numbers, including two variants of INCP-402, the same test kit relabeled by Biozek. The company also registered with Italy’s Ministry of Health as the manufacturer of the tests. Italian law allows for the buying and relabeling of imported tests as long as the importer carries out its own quality control and repackaging and discloses the original manufacturer if asked.
ScreenItalia declined to answer reporters’ questions about the original manufacturer of the tests, or where the tests were sold.
Also available in Italy, as well as many other countries, are tests sold by Acro Biotech, a company based in Rancho Cucamonga, California. The company sells tests globally under two brand names, Acro and JusCheck, which bear official variants of Alltest’s INCP-402 reference number. It also markets other rapid blood tests to detect drugs, infectious diseases, and markers of conditions like heart disease.
Filings with the U.S. Food and Drug Administration (FDA) show that Acro Biotech is a registered relabeler for Alltest. Such relabeling agreements are legal in the United States as long as they are properly declared with the FDA and a so-called Emergency Use Authorization is obtained. FDA records show that an application for such an authorization has been submitted for the test, but has not yet been granted.
Reporters did not uncover evidence that Acro Biotech has sold coronavirus test kits in the United States, but it has managed to find plenty of overseas buyers. Reporters found the tests for sale by distributors in India, Chile, Brazil, Spain, Italy, Belgium, and Finland.
Acro Biotech’s coronavirus tests are being sold in Europe with a CE mark that lists the manufacturer as the California-based company, according to a declaration of conformity posted online. (Reached by telephone, Acro Biotech CEO Joe Fan asked reporters to send questions by email, but did not respond.)
In Italy, one of the countries worst hit by COVID-19, the national government has shunned rapid antibody tests.
“Concerning the so-called ‘rapid test’ with a drop of blood, we do not recognize them as valid, and we stick to the international scientific community’s opinion on it,” Italian Ministry of Health spokesman Cesare Buquicchio said.
Many local authorities in Italy’s decentralized health system have bought such tests despite being “clearly advised against it,” Buquicchio said.
“I can assure you that some regions even had to throw them away because the tests aren’t working,” he said.
Despite this, Acro Biotech’s tests have been sold to local governments in at least three Italian regions. The local health service in the city of Cagliari bought 12,500 Acro Biotech kits and a hospital in Naples bought 2,500, according to tenders published by the two institutions.
Gianfranco Tedeschi, the mayor of Cerchio in the region of Abruzzo, also bought Acro Biotech rapid tests for each of the town’s 1,700 inhabitants in late March, believing they were made in California, despite the objections of the regional government. He said he was happy with the results and was not aware of any cases in which a false negative had caused problems.
“Our screening found infected people [who] were later confirmed with a swab test, but also antibodies in people who had already gone through the illness,” Tedeschi said.
“He Thought He Was Negative”
In much of the world, the problem has less to do with the tests themselves — which even skeptical scientists say are good for some things, such as community screening — than with sellers and clinics who inappropriately market and administer them.
Reporters found multiple instances of rapid tests being sold directly to customers by retailers online or operating via messaging platforms such as Telegram.
In some cases, it was not possible to trace how tests travelled from producers to sellers. In others, reporters were able to trace the role of intermediary companies and facilitators whose presence in the chain appears to have helped obscure the tests’ origins and contributed to their misuse.
In Macedonia, the test administered to Zekirovski had been supplied by a Swiss middleman, Spring Healthcare, that sold 500 Biozek-branded tests to the Acibadem Sistina Hospital for 8 euros each in mid-March, according to an invoice obtained by reporters. The tests came from a batch of about 100,000 sent by Inzek to Spring Healthcare, Inzek CEO Zeki Hamid told reporters.
In total, 50,000 Biozek tests were reportedly imported into North Macedonia, mostly via another middleman after the relationship between the Swiss and Dutch companies fell apart.
Wasay Bhatti, a consultant for Spring Healthcare, said that Inzek had misled his firm into believing the tests were made in the Netherlands.
“It was not clear that it’s fully manufactured by a Chinese company and that everything is done [in China],” Bhatti said.
“A lot of distributors have put that it’s been manufactured in the Netherlands, it’s got on the box it’s Netherlands,” he said. “Now we’ve told everybody it’s not the Netherlands anymore, it’s China.”
Adding to the confusion, Spring Healthcare for a time marketed Biozek’s tests to potential buyers under its own brand, according to promotional materials obtained by reporters from an online broker.
This was a temporary measure used only before signing on with customers, Bhatti said, in order to prevent them from cutting out the middleman and going straight to the Dutch company.
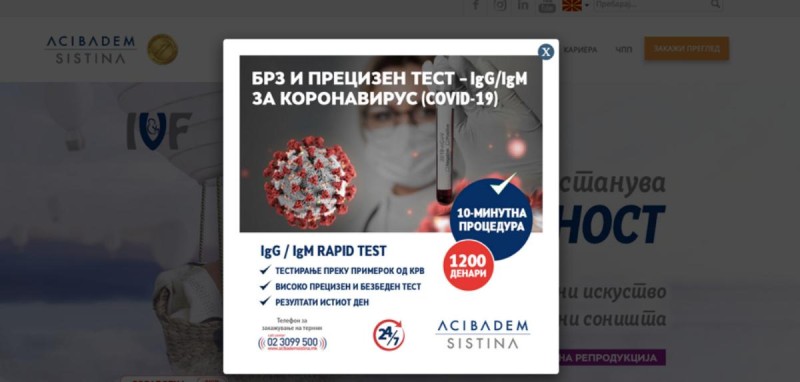
In North Macedonia, Biozek’s test were used as part of an aggressive marketing campaign by the Acibadem Sistina Hospital, which soon fell afoul of regulators. The “fast and efficient” tests were promoted online and through paid advertorials in local media.
In April, the country’s Agency for Medicines and Medical Devices (MALMED) ordered the hospital to take down all its advertisements for the tests, finding that the hospital had failed to adequately communicate the limited clinical usefulness of the tests to the public, agency head Visara Riza told OCCRP. The hospital had also advertised the tests without getting prior approval from MALMED, Visara said.
Zekirovski’s hospitalization shortly before his death severely affected the health system in his home city of Kumanovo. Once it was discovered that they had treated Zekirovski without taking the proper precautions, 72 medical workers had to be placed in isolation. Ten ultimately tested positive for the virus.
A false negative given by another Biozek test at Acibadem Sistina to a prominent journalist, Goce Mihajloski, resulted in the leaders of two of the country’s main political parties going into self-quarantine after they appeared alongside him on a television panel show.
“He thought he was negative. He was transparent and told us prior to the debate that he had symptoms and that he was tested negative in Sistina,” Zoran Zaev, the leader of North Macedonia’s Social Democrats, told OCCRP.
In response to questions, Acibadem Sistina Hospital said the tests were only being used for mass community screening. The hospital also stated, incorrectly, that the tests had been approved for use by the U.S. FDA.
The hospital said it was reporting journalists’ questions to the Dutch Embassy in North Macedonia “in order to protect a prominent producer from a partner country and good trade relations.”
Problems with the Biozek tests did not stop Acibadem Sistina from exporting them to neighboring Serbia. An invoice obtained by reporters shows the Macedonian hospital sold 300 of the kits for 10 euros each to Serbia’s Dedinje Institute of Cardiovascular Diseases, whose director is Milovan Bojic, a former health minister in the government of the late autocrat Slobodan Milosevic.
In response to questions from reporters, Bojic said the tests were sent as a donation and that the Serbian institute did not pay for them. It did, however, use the kits to test 14 patients.
Clarification: This story was changed to reflect the fact that, as of the time of publication, the World Health Organization recommended that rapid antibody tests for COVID-19 be used only for research purposes and not in any real-world settings.
Additional reporting by Lorenzo Bagnoli, Dragana Peco, Antonio Baquero, David Ilieski, Mariia Horban, Atanas Tchobanov, and Romana Puiulet.
This story was reported in collaboration with Bivol (Bulgaria), IRPIMedia (Italy), IRL Macedonia, KRIK (Serbia), Michelle Salomons and Karlijn Kuijpers of Investico (Netherlands), Important Stories (Russia), and Tempo (Indonesia).